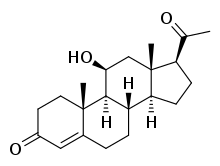
11β-Hydroxyprogesterone
 | |
| Names | |
|---|---|
| Preferred IUPAC name
(1S,3aS,3bS,9aR,9bS,10S,11aS)-1-Acetyl-10-hydroxy-9a,11a-dimethyl-1,2,3,3a,3b,4,5,8,9,9a,9b,10,11,11a-tetradecahydro-7H-cyclopenta[a]phenanthren-7-one | |
| Other names
11β-OHP; 21-Deoxycorticosterone; 21-Desoxycorticosterone; 11β-Hydroxypregn-4-ene-3,20-dione | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.009.088 |
| KEGG | |
PubChem CID |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
C21H30O3 |
| Molar mass | 330.468 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
11β-Hydroxyprogesterone (11β-OHP), also known as 21-deoxycorticosterone, as well as 11β-hydroxypregn-4-ene-3,20-dione, is a naturally occurring, endogenous steroid and derivative of progesterone.[1] It is a potent mineralocorticoid.[1] Syntheses of 11β-OHP from progesterone is catalyzed by the steroid 11β-hydroxylase (CYP11B1) enzyme,[2][3] and, to a lesser extent, by the aldosterone synthase enzyme (CYP11B2).[2]
Function
Along with its epimer 11α-hydroxyprogesterone (11α-OHP), 11β-OHP has been identified as a very potent competitive inhibitor of both isoforms (1 and 2) of 11β-hydroxysteroid dehydrogenase (11β-HSD).[4][5]
Outcome of 21-hydroxylase deficiency
It has been known since 1987 that increased levels of 11β-OHP occur in 21-hydroxylase deficiency.[6][7] A study in 2017 has shown that in subjects with 21-hydroxylase deficiency, serum 11β-OHP concentrations range from 0.012 to 3.37 ng/mL, while in control group it was below detection limit of 0.012 ng/mL.[8] 21-hydroxylase is an enzyme that is also involved in progesterone metabolism, producing 11-deoxycorticosterone. In normal conditions, 21-hydroxylase has higher activity on progesterone than steroid 11β-hydroxylase (CYP11B1) and aldosterone synthase (CYP11B2) that convert progesterone to 11β-OHP. That's why in 21-hydroxylase deficiency, given the normal function of the CYP11B enzymes, the progesterone is directed towards 11β-OHP pathway rather than towards 11-deoxycorticosterone pathway, that is also usually accompanied by an increase in progesterone levels.[9] In the normal route to aldosterone and cortisol, progesterone and 17α-hydroxyprogesterone are first hydroxylated at position 21 and then hydroxylated at other positions. In 21-hydroxylase deficiency, progesterone and 17α-hydroxyprogesterone accumulate and are the substrates of steroid 11β-hydroxylase, leading to 1β-OHP and 21-deoxycortisol, respectively.[10] In the 2017 study above mentioned, serum progesterone concentrations in boys (10 days to 18 years old) with 21-hydroxylase deficiency reached levels similar to female luteal values (up to 10.14 ng/mL, depending on severity and treatment), while in the control group of boys progesterone was 0.07 ng/mL (0.22 nmol/L) on average, ranged from 0.05 to 0.40 ng/mL.[8]
While studies suggest that 11β-OHP, also known as 21-deoxycorticosterone, can be used as marker for adrenal 21-hydroxylase deficiency,[6] another 21-carbon steroid — 21-deoxycortisol (produced from 17α-hydroxyprogesterone) gained acceptance for this purpose.[11][12]
See also
- 21-Deoxycortisol (11β,17α-dihydroxyprogesterone)
- 11-Deoxycorticosterone (21-hydroxyprogesterone)
- Corticosterone (11β,21-dihydroxyprogesterone)
- Cortisol (11β,17α,21-trihydroxyprogesterone)
- 11-Deoxycortisol (17α,21-dihydroxyprogesterone)
- 9α-Bromo-11-ketoprogesterone
References
- 1 2 "Human Metabolome Database: Showing metabocard for 11b-Hydroxyprogesterone (HMDB04031)". hmdb.ca. Retrieved 2016-12-16.
- 1 2 Strushkevich N, Gilep AA, Shen L, Arrowsmith CH, Edwards AM, Usanov SA, Park HW (February 2013). "Structural insights into aldosterone synthase substrate specificity and targeted inhibition". Molecular Endocrinology. 27 (2): 315–24. doi:10.1210/me.2012-1287. PMC 5417327. PMID 23322723.
- ↑ van Rooyen D, Gent R, Barnard L, Swart AC (April 2018). "The in vitro metabolism of 11β-hydroxyprogesterone and 11-ketoprogesterone to 11-ketodihydrotestosterone in the backdoor pathway". The Journal of Steroid Biochemistry and Molecular Biology. 178: 203–212. doi:10.1016/j.jsbmb.2017.12.014. PMID 29277707. S2CID 3700135.
- ↑ Souness GW, Latif SA, Laurenzo JL, Morris DJ (April 1995). "11 alpha- and 11 beta-hydroxyprogesterone, potent inhibitors of 11 beta-hydroxysteroid dehydrogenase (isoforms 1 and 2), confer marked mineralocorticoid activity on corticosterone in the ADX rat". Endocrinology. 136 (4): 1809–12. doi:10.1210/endo.136.4.7895695. PMID 7895695.
- ↑ Souness GW, Morris DJ (March 1996). "11 alpha- and 11 beta-hydroxyprogesterone, potent inhibitors of 11 beta-hydroxysteroid dehydrogenase, possess hypertensinogenic activity in the rat". Hypertension. 27 (3 Pt 1): 421–5. doi:10.1161/01.hyp.27.3.421. PMID 8698448.
- 1 2 Gueux B, Fiet J, Galons H, Boneté R, Villette JM, Vexiau P, et al. (January 1987). "The measurement of 11 beta-hydroxy-4-pregnene-3,20-dione (21-deoxycorticosterone) by radioimmunoassay in human plasma". primary. Journal of Steroid Biochemistry. 26 (1): 145–50. doi:10.1016/0022-4731(87)90043-4. PMID 3546944.
- ↑ Fiet J, Gueux B, Raux-DeMay MC, Kuttenn F, Vexiau P, Brerault JL, et al. (March 1989). "Increased plasma 21-deoxycorticosterone (21-DB) levels in late-onset adrenal 21-hydroxylase deficiency suggest a mild defect of the mineralocorticoid pathway". primary. The Journal of Clinical Endocrinology and Metabolism. 68 (3): 542–7. doi:10.1210/jcem-68-3-542. PMID 2537337.
- 1 2 Fiet J, Le Bouc Y, Guéchot J, Hélin N, Maubert MA, Farabos D, Lamazière A (March 2017). "A Liquid Chromatography/Tandem Mass Spectometry [sic] Profile of 16 Serum Steroids, Including 21-Deoxycortisol and 21-Deoxycorticosterone, for Management of Congenital Adrenal Hyperplasia". primary. Journal of the Endocrine Society. 1 (3): 186–201. doi:10.1210/js.2016-1048. PMC 5686660. PMID 29264476.
- ↑ Nie M, Cui MX, Mao JF, Tong AL, Chen S, Wang X, et al. (December 2016). "[Possibility of progesterone as the diagnostic biomarker of 21-hydroxylase deficiency]". Zhonghua Yi Xue Za Zhi. 96 (48): 3866–3869. doi:10.3760/cma.j.issn.0376-2491.2016.48.003. PMID 28057154.
- ↑ Turcu AF, Auchus RJ (June 2015). "Adrenal steroidogenesis and congenital adrenal hyperplasia". Endocrinology and Metabolism Clinics of North America. 44 (2): 275–96. doi:10.1016/j.ecl.2015.02.002. PMC 4506691. PMID 26038201.
- ↑ Cristoni S, Cuccato D, Sciannamblo M, Bernardi LR, Biunno I, Gerthoux P, et al. (2004). "Analysis of 21-deoxycortisol, a marker of congenital adrenal hyperplasia, in blood by atmospheric pressure chemical ionization and electrospray ionization using multiple reaction monitoring". primary. Rapid Communications in Mass Spectrometry. 18 (1): 77–82. Bibcode:2004RCMS...18...77C. doi:10.1002/rcm.1284. PMID 14689562.
- ↑ Sarathi V, Atluri S, Pradeep TV, Rallapalli SS, Rakesh CV, Sunanda T, Kumar KD (2019). "Utility of a Commercially Available Blood Steroid Profile in Endocrine Practice". primary. Indian Journal of Endocrinology and Metabolism. 23 (1): 97–101. doi:10.4103/ijem.IJEM_531_18. PMC 6446682. PMID 31016162.