Axelopran
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
IUPAC name
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
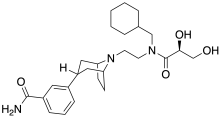
| Formula | C26H39N3O4 |
| Molar mass | 457.615 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Axelopran (INN, USAN) (developmental code name TD-1211) is a drug which is under development by Theravance Biopharma for the treatment of opioid-induced constipation. It acts as a peripherally acting μ-opioid receptor antagonist and also acts on κ-, and δ-opioid receptors, with similar affinity for the μ- and κ-opioid receptors and about an order of magnitude lower affinity for the δ-opioid receptor. Axelopran has potent μ-opioid receptor antagonist activity on the gastrointestinal tract in vivo, and thus it produces a dose-dependent inhibition of opioid-induced delaying in gastric emptying in mice and rats following subcutaneous or oral administration.[1][2]
See also
References
- ↑ Armstrong SR, Campbell CB, Richardson CL, Vickery RG, Tsuruda PR, Long DD, et al. (June 2013). "The in vivo pharmacodynamics of the novel opioid receptor antagonist, TD-1211, in models of opioid-induced gastrointestinal and CNS activity". Naunyn-Schmiedeberg's Archives of Pharmacology. 386 (6): 471–8. doi:10.1007/s00210-013-0844-5. PMID 23512167. S2CID 15482326.
- ↑ Tsuruda PR, Vickery RG, Long DD, Armstrong SR, Beattie DT (June 2013). "The in vitro pharmacological profile of TD-1211, a neutral opioid receptor antagonist". Naunyn-Schmiedeberg's Archives of Pharmacology. 386 (6): 479–91. doi:10.1007/s00210-013-0850-7. PMID 23549670. S2CID 18963203.
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.