Volinanserin
 | |
| Clinical data | |
|---|---|
| Other names | MDL-100,907; M100907 |
| ATC code |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.123.797 |
| Chemical and physical data | |
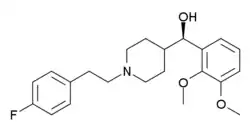
| Formula | C22H28FNO3 |
| Molar mass | 373.468 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
Volinanserin (INN) (developmental code name MDL-100,907) is a highly selective 5-HT2A receptor antagonist that is frequently used in scientific research to investigate the function of the 5-HT2A receptor.[1][2][3] It was also tested in clinical trials as a potential antipsychotic,[4][5] antidepressant,[6] and treatment for insomnia but was never marketed.[7]
See also
References
- ↑ Schmidt CJ, Fadayel GM, Sullivan CK, Taylor VL (November 1992). "5-HT2 receptors exert a state-dependent regulation of dopaminergic function: studies with MDL 100,907 and the amphetamine analogue, 3,4-methylenedioxymethamphetamine". European Journal of Pharmacology. 223 (1): 65–74. doi:10.1016/0014-2999(92)90819-P. PMID 1362159.
- ↑ Herth MM, Kramer V, Piel M, Palner M, Riss PJ, Knudsen GM, Rösch F (April 2009). "Synthesis and in vitro affinities of various MDL 100907 derivatives as potential 18F-radioligands for 5-HT2A receptor imaging with PET". Bioorganic & Medicinal Chemistry. 17 (8): 2989–3002. CiteSeerX 10.1.1.519.5663. doi:10.1016/j.bmc.2009.03.021. PMID 19329329.
- ↑ Nic Dhonnchadha BA, Fox RG, Stutz SJ, Rice KC, Cunningham KA (April 2009). "Blockade of the serotonin 5-HT2A receptor suppresses cue-evoked reinstatement of cocaine-seeking behavior in a rat self-administration model". Behavioral Neuroscience. 123 (2): 382–96. doi:10.1037/a0014592. PMC 3830454. PMID 19331461.
- ↑ Offord SJ, Wong DF, Nyberg S (August 1999). "The role of positron emission tomography in the drug development of M100907, a putative antipsychotic with a novel mechanism of action". Journal of Clinical Pharmacology. 39 (S1): 17S–24S. doi:10.1002/j.1552-4604.1999.tb05933.x. PMID 10434243. S2CID 21311671.
- ↑ Charney DS, Nestler PS, Sklar P, Buxbaum JD (July 2013). Neurobiology of Mental Illness. OUP USA. p. 767. ISBN 9780199934959.
- ↑ Marek GJ, Martin-Ruiz R, Abo A, Artigas F (December 2005). "The selective 5-HT2A receptor antagonist M100907 enhances antidepressant-like behavioral effects of the SSRI fluoxetine". Neuropsychopharmacology. 30 (12): 2205–15. doi:10.1038/sj.npp.1300762. PMID 15886717.
- ↑ Teegarden BR, Al Shamma H, Xiong Y (2008). "5-HT(2A) inverse-agonists for the treatment of insomnia". Current Topics in Medicinal Chemistry. 8 (11): 969–76. doi:10.2174/156802608784936700. PMID 18673166.
| Typical |
|
|---|---|
| Disputed | |
| Atypical |
|
| Others |
|
| |
| GABAA |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| GABAB | |||||||||||||||||||||||||
| H1 |
| ||||||||||||||||||||||||
| α2-Adrenergic |
| ||||||||||||||||||||||||
| 5-HT2A |
| ||||||||||||||||||||||||
| Melatonin | |||||||||||||||||||||||||
| Orexin | |||||||||||||||||||||||||
| α2δ VDCC | |||||||||||||||||||||||||
| Others |
| ||||||||||||||||||||||||
Serotonin receptor modulators | |||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-HT1 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT2 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT3–7 |
| ||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.