Gevotroline
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
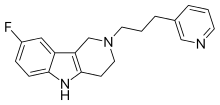
| Formula | C19H20FN3 |
| Molar mass | 309.388 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
Gevotroline (WY-47,384) is an atypical antipsychotic with a tricyclic structure which was under development for the treatment of schizophrenia by Wyeth-Ayerst.[1][2][3] It acts as a balanced, modest affinity D2 and 5-HT2 receptor antagonist and also possesses high affinity for the sigma receptor.[2][4][5][6] It was well tolerated and showed efficacy in phase II clinical trials but was never marketed.[2][3]
See also
References
- ↑ David J. Triggle (1996). Dictionary of Pharmacological Agents. Boca Raton: Chapman & Hall/CRC. ISBN 0-412-46630-9.
- 1 2 3 Bristol, James A. (1990). Annual Reports in Medicinal Chemistry (Volume 25). Boston: Academic Press. ISBN 0-12-040525-3.
- 1 2 Stone, T. W.; Stone, Trevor (1996). CNS neurotransmitters and neuromodulators: dopamine. Boca Raton: CRC Press. ISBN 0-8493-7632-7.
- ↑ Snyder SH, Largent BL (1989). "Receptor mechanisms in antipsychotic drug action: focus on sigma receptors". The Journal of Neuropsychiatry and Clinical Neurosciences. 1 (1): 7–15. doi:10.1176/jnp.1.1.7. PMID 2577720.
- ↑ Matheson GK, Guthrie D, Bauer C, Knowles A, White G, Ruston C (January 1991). "Sigma receptor ligands alter concentrations of corticosterone in plasma in the rat". Neuropharmacology. 30 (1): 79–87. doi:10.1016/0028-3908(91)90046-E. PMID 1675451. S2CID 29702968.
- ↑ Gudelsky GA, Nash JF (February 1992). "Neuroendocrinological and neurochemical effects of sigma ligands". Neuropharmacology. 31 (2): 157–62. doi:10.1016/0028-3908(92)90026-L. PMID 1348112. S2CID 36585024.
| Typical |
|
|---|---|
| Disputed | |
| Atypical |
|
| Others |
|
| |
| Classes |
|
|---|---|
| Antidepressants (TCAs and TeCAs) |
|
| Antihistamines |
|
| Antipsychotics |
|
| Anticonvulsants | |
| Others |
|
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.